The promising battery solutions that make use of metallic lithium as the anode are lithium metal batteries. They use a single layer of lithium close to the anode rather than electrode materials that store lithium ions, as is the case with most lithium-ion batteries (LiBs), which can significantly reduce their size and weight.
While these batteries could outflank LiBs concerning limit and energy thickness, to work dependably, the layer between their Li metal anode and electrolyte ought to be powerful and stable over the long haul. The solid electrolyte interphase (SEI) layer has been difficult to stabilize up to this point.
A new method recently developed by Stanford University researchers has the potential to produce a more stable SEI in lithium metal batteries, thereby enhancing their overall cycling performance. A salt- and solvent-phobic polymer is used to coat the battery cell’s electrodes in their proposed method, which was presented in a Nature Energy paper.
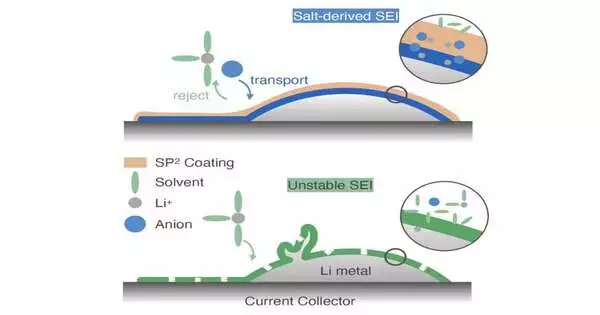
“The reaction between Li and the electrolyte is the cause of Li metal battery capacity fading. Li, in particular, combines with the salt and solvents to generate an SEI layer. According to recent study, an SEI produced from Li reacting with salt (salt-derived SEI) is more stable than a solvent-derived SEI and is associated with a longer cell cycle life.”
Rachel Z Huang, one of the researchers who carried out the study,
One of the researchers who carried out the study, Rachel Z. Huang, told Tech Xplore, “The reaction between Li and the electrolyte is the origin of Li metal battery capacity fading.” In particular, an SEI layer is produced when Li interacts with solvents and salt. A salt-derived SEI, which is derived from Li reacting with salt, is associated with a longer cell cycle life and is more stable than solvent-derived SEI, according to recent research.
Huang and her colleagues decided to cover the electrodes with a polymer coating that had been carefully designed in order to cause salt-derived SEI formation in lithium metal batteries. The polymer they made comprises a polysiloxane spine with pyridinium bis(trifluoromethylsulfonyl)imide (PyTFSI) and perfluorinated side chains.
“Our point was for the polymer to at the same time expand the presence of salt and abate the presence of dissolvable at the connection point, in this way driving salt-determined SEI development,” Huang made sense of. “We have planned a siloxane-based polymer with fastened salt-philic and dissolvable phobic (SP2) side chains. In an H-Cell experiment, this polymer increased the salt-derived SEI content with three main types of electrolytes and demonstrated selective salt transport over solvent.”
H-cells explore showing SP2 just permits salt that is not dissolvable to go through. Credit: Rachel Z. Huang
The detailing of the polymer covering utilized by Huang and her associates was recognized in the wake of investigating a few potential side chain sciences. It was determined to be the best solution during their initial analyses, both in terms of its electrochemical performance and its interaction with salt and solvent.
In a series of experiments, the researchers looked at their plan and found that it could control the SEI in lithium metal battery cells, which would make them last longer. They also tried out their polymer-coating method with a variety of electrolytes, such as ether (lithium bis(trifluoromethanesulfonyl)imide in 1,3-dioxolane/1,2-dimethoxyethane with 1 weight percent LiNO3), carbonate (lithium hexafluorophosphate in ethylene carbonate/diethyl carbonate with 10% fluoroethylene carbonate), and fluorinated 1,4
Huang stated, “We achieved one of the state-of-the-art cycling performances for Li metal batteries by utilizing physics to tune chemistry in this work.” The critical highlights of our methodology are its adaptability in plan and flexibility in application. We demonstrated that our interface design can be modulated, which means that different polymer side chains and/or backbones can be used to achieve design flexibility. Additionally, composites can be applied to this modulated design concept, providing even more design options.”
So far, the researchers have demonstrated that their polymer coating works well with three main types of electrons found in lithium metal batteries. Notwithstanding, while additional promising electrolytes are revealed, the arrangement of electrolytes that answer well to their strategy could be expanded further.
As it seems to sum up well across cells with different organizations, this promising plan system could before long be utilized by different groups overall to work on the exhibition and dependability of lithium metal batteries. In addition, Huang and her colleagues intend to conduct additional studies to evaluate the benefits and applicability of their method.
“Our approach offers broader applicability and addresses multiple electrolyte systems, whereas other works that aim to increase interfacial stability typically focus on a specific electrolyte,” Huang added. In order to find out what other compositions are capable of achieving these salt-phobic and solvent-philic properties as well as the potential of this design when paired with novel electrolyte formulations, we intend to conduct additional research into the materials design space.”
More information: Zhuojun Huang et al, A salt-philic, solvent-phobic interfacial coating design for lithium metal electrodes, Nature Energy (2023). DOI: 10.1038/s41560-023-01252-5