Another dengue treatment that could turn into the first to forestall and treat the infection has demonstrated success in beginning preliminary tests in monkeys, as per a new examination.
Dengue is transmitted by mosquitoes and influences several million people every year, delivering ruthless side effects that deserve the moniker “breakbone fever”.
It is endemic in many nations, yet no treatment exists, and two antibodies that have been created are not yet universally endorsed.
A long time ago, specialists distributed work showing a compound could really keep the infection from duplicating in cell societies and mice by forestalling the connection between two proteins.
“The molecule has now been developed and tested in mice and monkeys, with extremely encouraging findings.”
Marnix Van Loock, lead for emerging pathogens at the Janssen Companies of Johnson & Johnson,
Presently, the group has refined the compound and tried it in two mice and monkeys, with “extremely reassuring” results, said Marnix Van Loock, lead for arising microbes at the Janssen Organizations of Johnson & Johnson, a medication organization.
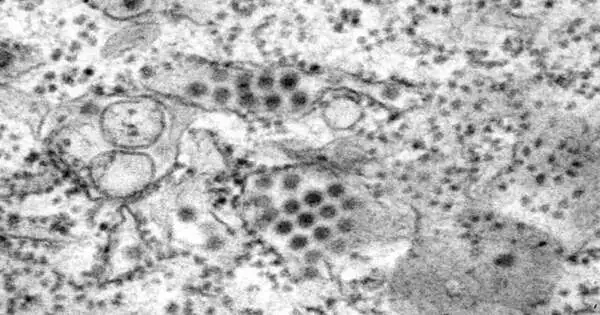
In rhesus macaques, a high portion of the compound known as JNJ-1802 “totally hindered viral replication”, he told AFP, while in control creatures viral RNA was identified between days three and seven after disease.
In monkeys, the compound was tested against the two most common of the four kinds of dengue, just for its safeguarding properties as opposed to for treatment.
Yet, it was tried for both treatment and avoidance in mice against every one of the four kinds of dengue, with fruitful results, Van Loock said.
Dengue can cause extraordinary influenza-like side effects and, at times, forms into a serious structure that can be lethal.
Since there are four unique strains, getting tainted by one doesn’t safeguard against another, and getting dengue a second time is in many cases more serious.
Scientists have cautioned that a hotter, wetter environment that is more friendly to mosquitoes is probably going to increase the frequency of infections passed on by the bug.
With no treatment available, endeavors right now center around lessening transmission by incorporating tainting mosquitoes with microorganisms.
An immunization called Dengvaxia is endorsed for use only in certain nations and is successful against a single strain.
A subsequent immunization, Qdenga, was supported last December for use by the European Association, and it has likewise been greenlighted by England and Indonesia.
There are still inquiries to address about the treatment in any case, including whether it could expand weakness to reinfection.
At the point when individuals contract dengue, the presence of the infection in their blood for the most part animates a powerful immune reaction that shields them from future contamination.
Yet, in certain individuals, the safe reaction is more vulnerable, which leaves them powerless against reinfection, which can deliver more serious side effects.
It isn’t yet evident whether forestalling or diminishing viral replication could create that equivalent weakness to reinfection.
The scientists should submit wellbeing information from their ongoing period of testing prior to pushing forward with additional preliminary steps, including people and field reads for regions impacted by dengue.
Van Loock was hesitant to conjecture on when a treatment could be practically deployed.
“We are directed by the science and the information that we produce to truly respond to that inquiry,” he said.