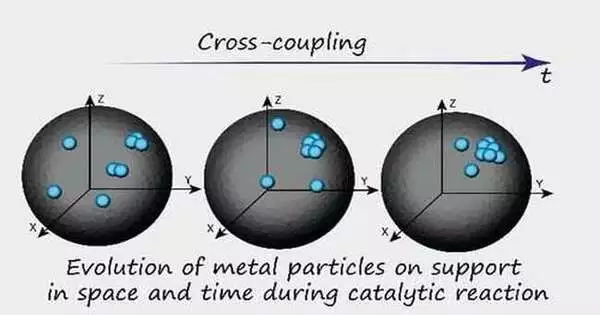
A small group of chemists at the Russian Academy of Sciences has discovered that catalysts used in fine organic synthesis rely heavily on metal atoms rather than nanoparticles. In the study, which was published in the Journal of the American Chemical Society, the team tracked a region of a catalyst during a reaction using a variety of electron microscopy techniques to learn more about the process.
There are two primary approaches to studying a reaction, as previous research has demonstrated. The most fundamental is the first: the reaction is simply observed and/or measured as new ingredients are added. The use of high-speed cameras can make this easier. Nanoscale reactions, of course, will not benefit from this strategy. In such instances, chemists employ a second strategy: they endeavor to catch the condition of the multitude of parts when the response occurs and afterward contrast them with looking further into what occurred.
However, there is no way to demonstrate that the studied objects correspond to one another, so this second method fails miserably. Lately, scientists have been dealing with another methodology: following the activity of a solitary molecule during the response. This new method has been shown to be useful, but it also has some drawbacks: it can’t be used for reactions that happen in the nanoworld. The researchers combined machine-learning algorithms with a variety of electron microscopy techniques in this new endeavor.
The researchers used a carbon substrate with palladium nanoparticles embedded as catalysts to test their theories. They were able to follow a portion of the catalyst as it progressed through a reaction by employing a variety of electron microscopes to examine the reactions that were carried out with this catalyst and then using the results to train a machine learning algorithm. In addition to the nanoparticles, they could see that there were clusters and individual metal atoms involved in the reaction. Even though nanoparticles only make up 1% of the palladium’s mass, subsequent research revealed that the palladium atoms were responsible for approximately 99% of the catalytic activity.
More information: Alexey S. Galushko et al, Time-Resolved Formation and Operation Maps of Pd Catalysts Suggest a Key Role of Single Atom Centers in Cross-Coupling, Journal of the American Chemical Society (2023). DOI: 10.1021/jacs.3c00645