According to mouse cell research, correcting DNA disorganization in mouse cells may aid in the diagnosis and treatment of rare inherited diseases. Researchers say they discovered that a protein that helps form a structural network under the surface of the cell’s ‘command center,’ its nucleus, is critical to ensuring that DNA inside it remains orderly in a study with lab-grown mouse cells.
In a study with lab-grown mouse cells, Johns Hopkins Medicine researchers discovered that a protein that aids in the formation of a structural network beneath the surface of the cell’s “command center” its nucleus is critical to ensuring that the DNA inside it remains orderly. The new experiments distinguish the role of the protein, known as lamin C, revealing its utility in diagnosing and treating a variety of genetic disorders linked to DNA disorganization, such as progeria, muscular dystrophy, and heart disorders caused by mutations in these and related proteins.
“Because lamin C appears to be important for genome organization in general, the implications of these findings may go beyond the known laminopathic diseases. We simply do not know how lamin C behaves in other diseases characterized by genome dysregulation at this time “Karen Reddy, Ph.D., an assistant professor of biological chemistry at Johns Hopkins University School of Medicine, agrees.
She goes on, “Many people are aware that inherited diseases are caused by gene mutations, or errors in the genetic code. However, highly disorganized genes may be just as effective as mutations in causing disease.”
Reddy observes that most genetic tests do not take into account the mechanics of how DNA is organized, which may be an important foundation for understanding genetic diseases. Reddy and her colleagues published the findings of their study in Genome Biology.
Many people are aware that inherited diseases are caused by gene mutations, or errors in the genetic code. However, highly disorganized genes may be just as effective as mutations in causing disease.
Karen Reddy
The nucleus of each human cell contains approximately 6 feet of tightly coiled DNA that contains the genetic instructions for every structure and function in the body. These DNA threads must be organized into usable parts in order for the cell to function. The lamin proteins, which attach to the surface of the nucleus, accomplish this by grabbing onto DNA segments and keeping them separate and tidy.
“Each compartment created by a lamin acts like a kitchen utensil drawer, keeping knives, forks, and spoons easily accessible while keeping less frequently used items like serving pieces out of the way until needed,” Reddy explains.
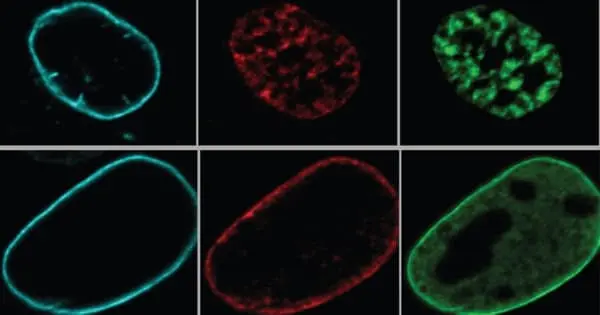
Reddy and her colleagues used fluorescent dyes to track three types of lamin proteins A, B, and C through cell division, when DNA from one cell is duplicated and split between two offspring cells, in order to better understand how lamins influence how the cell uses and organizes its DNA.
While lamin B has been easily distinguished in previous studies, lamin A and C have traditionally been treated as duplicate proteins because they are produced by the same gene, according to Reddy. However, there was mounting evidence that A and C type lamins served distinct functions.
To distinguish between them, Reddy’s team genetically modified mouse embryonic cells to remove either the gene that produces lamin B or the gene that contains both lamins A and C. The researchers then used microscopes to observe how the lamins behaved and whether the nuclear DNA of the cells remained organized as it divided.
The researchers discovered that nuclear DNA in cells lacking lamin B looked nearly identical to normal dividing cells, implying that lamin B may not be required for DNA reorganization after cell division. Nuclear DNA in cells lacking lamins A and C, on the other hand, did not reorganize neatly, becoming tangled and disorganized from its normal compartments within the nucleus.
“It looked like a rowdy party was going on in the normally well-organized kitchen,” Reddy says of the cells lacking lamins A and C. “Things were not in their proper places, and the strands of active and inactive DNA were mixed together and separated from the lamins at the nucleus’s edge.”
The researchers then used a series of specialized chemical reagents to disable either lamin A or lamin C in mouse cells, allowing them to test each protein separately. Cells lacking lamin A appeared to be able to reorganize after cell division just as well as normal cells. However, in cells lacking lamin C, nuclear DNA organization became disorganized once more.
According to Reddy, the behavior of lamin C in dividing cells revealed the reason for this distinction. While lamins A and B quickly bind to the surface of a newly formed nucleus and begin grabbing sections of DNA, lamin C remains dispersed throughout the nucleus and retains a special molecular tag known as phosphorylation. The researchers believe that this modified lamin C aids in the placement of DNA during reorganization. Once the DNA is organized, lamin C loses its molecular tag and joins the rest of the lamins at the nucleus’s edge.
“There is this exquisite choreography of the different lamin proteins and DNA to get things just right,” Reddy explains. The findings suggest that new tests that distinguish between lamins A and C could be developed and should be considered when screening for some genetic diseases that involve lamin proteins or other proteins at the nucleus’s edge.
The gene that codes for lamins A and C is linked to three types of muscular dystrophy: familial partial lipodystrophy, a condition that causes abnormal fatty tissue distribution; progeria; and several heart muscle disorders.
The findings raise several new questions, according to the researchers, including the role of lamins in organizing and regulating DNA during development. Because there appears to be some cross-talk between the different types of lamins, the team hopes to identify how lamin proteins and the genome behave when one specific type of lamin is mutated or disrupted. They also intend to investigate the cellular pathways that control the lamin proteins, particularly lamin C, in order to better understand the significance of its role in DNA control.