Correlative light electron microscopy (CLEM) is a powerful tool for bioimaging because it combines electron microscopy’s high spatial resolution and ultrastructural information with light microscopy’s ability to image living cells over large fields of view with molecular specificity. Researchers must ensure that biomolecules of interest are labeled with probes that are visible in both LM (typically by fluorescence) and EM (using electron-dense material) in order to accurately highlight and position them in CLEM. However, there are a number of problems with the probes that are currently available, such as their lack of integrity and stability under LM (photobleaching).
A group of researchers, led by Professor Paola Borri from Cardiff University and Professor Paul Verkade from the University of Bristol in the United Kingdom, have developed a brand-new CLEM method that does not require the use of fluorescent probes and instead makes use of small gold nanoparticles as a single probe that is visible in both LM and EM and has high contrast and photostability.
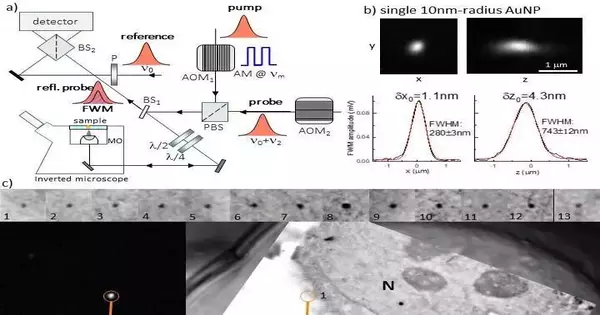
They used an optical microscope made by Professor Borri’s group for LM. This magnifying instrument distinguishes the non-direct optical reaction of gold nanoparticles, called four-wave blending (FWM). The researchers were able to locate individual small gold nanoparticles as single probes of the epidermal growth factor protein in human cancer cells using this strategy. These gold nanoparticles were located with nanometric precision and were free of background using light microscopy. They were also correlatively mapped to the associated transmission electron microscopy image with high accuracy.
FWM-CLEM permits highly accurate correlative microscopy workflows without the use of unstable fluorophores or additional fiducial markers thanks to the background-free and photostable FWM response of individual gold nanoparticles, which is well visible in electron microscopy due to their electron dense composition. This greatly simplifies sample preparation protocols.
The following is a summary provided by the researchers of the FWM microscope’s operational principle: A fourth wave is produced when three light fields interact with a medium to form FWM, a third-order non-linear light-matter interaction phenomenon. In this case, we employ a method in which the optical absorption peak of a gold nanoparticle is in resonance with all light waves having the same center frequency. For excitation and location, we utilize a mix of short optical beats of around 150 fs, called siphon and test, created by a similar laser source. The probe beam that is scattered by the gold nanoparticle in the presence of the pump changes as a result of the pump-induced change in the gold nanoparticle’s ability to transmit and scatter light.
“FWM detection is extremely high contrast and photo-stable. With this technique, we can identify individual little (down to 5nm range) gold nanoparticles inside dissipating and autofluorescing cells and tissues totally liberated from foundation, at imaging paces and excitation abilities viable with live cell imaging, with a responsiveness restricted exclusively by photon-shot commotion. We have likewise seen that FWM is a delicate columnist of gold nanoparticle shapes, promising for multiplexing by shape recognition in ongoing applications.”
“FWM-CLEM opens new exciting possibilities for correlative light electron microscopy workflows, overcoming existing limitations with fluorescent probes and/or fiducial markers that are not required in our method,” the researchers conclude. In general, we think that FWM-CLEM will have a big effect. FWM, for instance, opens the exciting possibility of tracking single viruses over extended observation times, background-free, deep within living cells and tissues, and then using CLEM to pinpoint events of interest (such as genome release) in the context of the cellular ultrastructure.” This is possible when combined with existing methods to label virions with gold nanoparticles or even encapsulate them inside virions.
More information: Iestyn Pope et al, Correlative light-electron microscopy using small gold nanoparticles as single probes, Light: Science & Applications (2023). DOI: 10.1038/s41377-023-01115-4