Organic molecules that have just been created can be tuned to emit specific colors based on their crystallized molecular structures.
Chemicals with molecular structures known as molecular switches have the ability to switch between two or more stable configurations in response to environmental changes. In the creation of molecular machines, computers, and drug delivery systems, they have a keen interest. Compounds with conformational isomers, which have the same molecular formula but different molecular structures, can function as very efficient molecular switches.
An approach to creating potential molecular switches from anthraquinodimethanes (AQDs), a class of crowded organic molecules, has been developed by researchers at Hokkaido University and Kyushu University. The research, which was led by associate professors Yusuke Ishigaki of Hokkaido University and Toshikazu Ono of Kyushu University, was published in the journal Materials Chemistry Frontiers.
“There are two common isomers, folded and twisted.” They are particularly fascinating as molecular switches because their sterically hindered double bond may create isomers that absorb and emit light at various wavelengths.”
Professor Toshikazu Ono at Kyushu University
According to Ono, “AQDs are a type of overcrowded ethylene molecule with carbon-carbon double bonds surrounded by large chemical groups.”. They exist in folded and twisted forms, which are their two main isomers. Since their sterically hindered double bond can produce isomers absorbing and emitting light at various wavelengths, they are particularly intriguing as molecular switches.”.
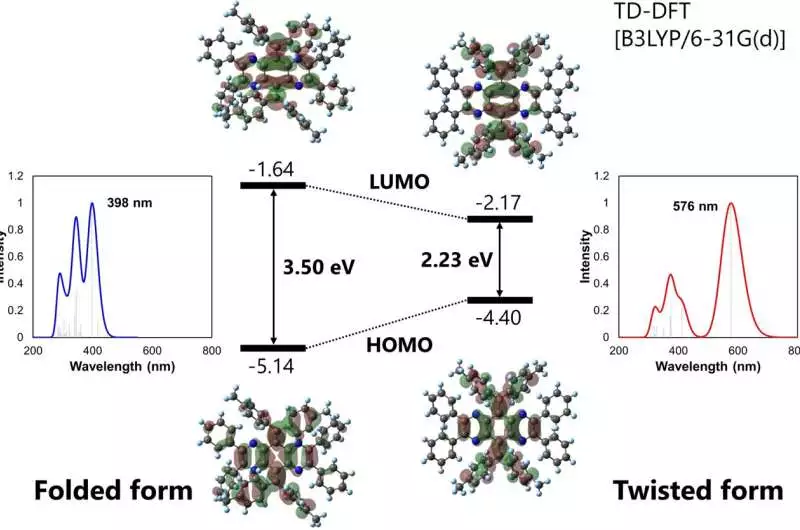
Different light wavelengths are absorbed by the folded and twisted isomers. Credit: Materials Chemistry Frontiers (2023). DOI: 10.1039/D2QM01199A
It is challenging to obtain pure samples of any other isomer to investigate its properties because AQDs typically adopt the most stable folded or twisted form. The researchers overcame this difficulty by creating flexible AQD derivatives that form various isomers more readily and steadily.
When recrystallized in various solvents, the synthesized derivatives were able to form other isomeric forms in addition to the stable twisted and folded isomers. To fully comprehend the derivatives’ properties, the researchers conducted a thorough analysis.
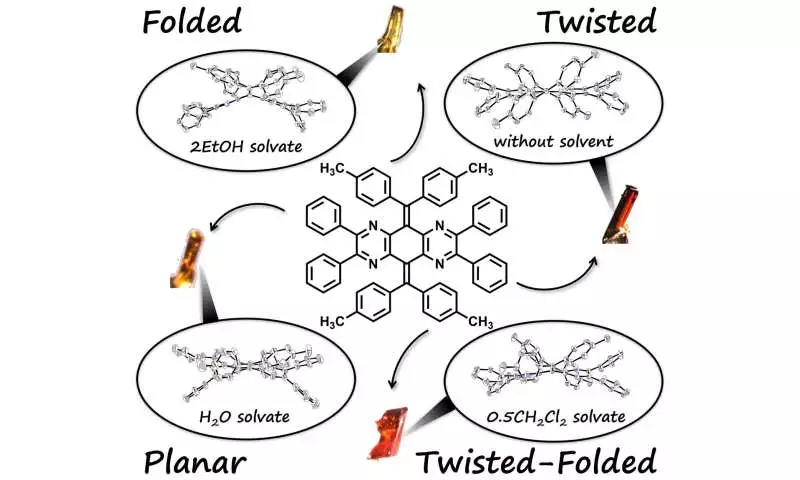
Credit: Materials Chemistry Frontiers (2023). DOI: 10.1039/D2QM01199A
Each of these isomers absorbs and emits a particular frequency of light when they are crystallized, which is caused by variations in the distribution of electrons within the isomer molecules. Intriguingly, when the crystals were ground into an amorphous solid, the light absorption and emission changed, and after treatment with the proper solvents, original or other crystals with a variety of colors can be produced.

Credit: Materials Chemistry Frontiers (2023). DOI: 10.1039/D2QM01199A
Ishigaki declared, “This work is the first report on the isolation of multiple isomeric forms of AQD. These compounds are excellent candidates for the creation of molecular switches because they can absorb and emit light at various frequencies and, more importantly, because they can modulate their absorption and emission in response to outside stimuli.”.
More information: Kazuma Sugawara et al, Exceptionally flexible quinodimethanes with multiple conformations: polymorph-dependent colour tone and emission of crystals, Materials Chemistry Frontiers (2023). DOI: 10.1039/D2QM01199A





