A new nanoparticle sensor developed by engineers at MIT has the potential to enable a straightforward urine test for the early detection of cancer. The sensors, which can recognize various malignant proteins, could likewise be utilized to recognize the kind of growth or the way things are responding to treatment.
When the nanoparticles come into contact with a tumor, they are designed to release brief DNA sequences that are excreted in the urine. These DNA “barcodes” can be analyzed to reveal specific characteristics of a patient’s tumor. The specialists planned their test with the goal that it very well may be performed utilizing a portion of paper, like an at-home coronavirus test, which they trust could make it reasonable and open to however many patients would be prudent.
“We are attempting to develop in a setting that makes innovation accessible to low- and middle-income settings. Sangeeta Bhatia, the John and Dorothy Wilson Professor of Health Sciences and Technology at MIT and a member of the Koch Institute for Integrative Cancer Research and Institute for Medical Engineering and Science at MIT, states, “Putting this diagnostic on paper is part of our goal of democratizing diagnostics and creating inexpensive technologies that can give you a quick answer at the point of care.”
“We are attempting to innovate by bringing technology to low- and middle-resource environments. Our mission is to democratize diagnoses and develop low-cost technologies that can provide you with an immediate response at the point of treatment, and part of that goal is to put this diagnostic on paper.”
Sangeeta Bhatia, the John and Dorothy Wilson Professor of Health Sciences and Technology
The researchers demonstrated, through experiments on mice, that they were able to employ the sensors to detect the activity of five distinct enzymes that are expressed in tumors. Using a microfluidic device to analyze the samples, they also demonstrated that their method could be scaled up to distinguish at least 46 distinct DNA barcodes in a single sample.
The paper, which was published today in Nature Nanotechnology, was led by Bhatia. The study’s lead author is Liangliang Hao, a former research scientist at MIT who is now an assistant professor of biomedical engineering at Boston University.
DNA barcodes
Bhatia’s lab has been working on “synthetic biomarkers” that could be used to diagnose cancer for several years. The idea behind this work is to find cancer biomarkers in a patient’s blood, like proteins or circulating tumor cells. Although synthetic biomarkers can be used to amplify smaller-scale changes that occur within small tumors, these naturally occurring biomarkers are so rare that it is nearly impossible to find them, particularly at an early stage.
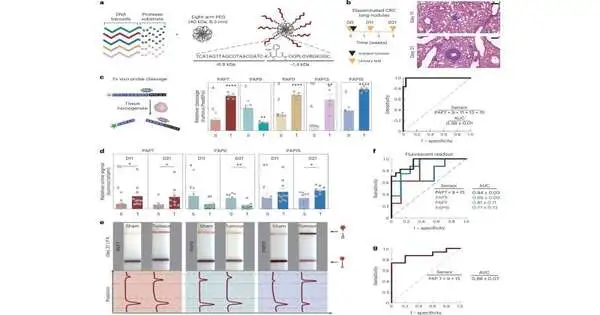
Bhatia has previously developed nanoparticles that are able to detect the activity of proteases, enzymes that aid cancer cells in escaping or settling into new environments by cutting through extracellular matrix proteins. Peptides that are cleaved by a variety of proteases are coated on the nanoparticles. After these peptides are released into the bloodstream, they can be concentrated and more easily detected in a urine sample.
A mass spectrometer was used to identify the initial peptide biomarkers based on minutely altered mass variations. Since this kind of equipment might not be available in low-resource settings, the researchers set out to create sensors that could be analyzed with DNA barcodes that can be read using CRISPR technology in a way that is easier and less expensive.
For this way to deal with work, the specialists needed to utilize a substance change called phosphorothioate to shield the circling DNA columnist scanner tags from being separated in the blood. This change has already been used to make modern RNA vaccines more stable, allowing them to stay in the body for longer.
Each DNA barcode is linked to a nanoparticle by a linker that can be cleaved by a specific protease, just like the peptide reporters. The DNA molecule is let go and is free to move around, eventually ending up in the urine if that protease is present. The researchers used two distinct kinds of nanoparticles for this study: one is a particle made of polymers that has been approved by the FDA for use in humans. The other is a “nanobody,” which is a fragment of an antibody that can be made to accumulate at the site of a tumor.
A paper strip that recognizes a reporter that is activated by a CRISPR enzyme called Cas12a can be used to analyze the sample once the sensors have been secreted in the urine. Cas12a amplifies the signal to produce a dark strip on a paper test when a particular DNA barcode is present in the sample.
The particles can be intended to convey a wide range of DNA-standardized tags, every one of which recognizes an alternate kind of protease movement, which is considered “multiplexed” detection. The test can more easily distinguish between different types of tumors thanks to an increase in both sensitivity and specificity brought about by using a larger number of sensors.
Disease signatures
The researchers demonstrated, through experiments on mice, that a panel of five DNA barcodes could accurately differentiate between tumors that first developed in the lungs and tumors created by colorectal cancer cells that had spread to the lungs.
According to Hao, “our goal here is to build up disease signatures and to see if we can use these barcoded panels not only to read out a disease but also to classify a disease or differentiate different types of cancer.”
Due to the wide range of tumors found in patients, the researchers anticipate that they will need to employ more than five barcodes for use in humans. A microfluidic chip that can read up to 46 different DNA barcodes from a single sample was developed in collaboration with researchers at the Broad Institute of MIT and Harvard, led by Harvard University Professor Pardis Sabeti.
This sort of testing could be utilized for recognizing disease, but additionally for estimating how well a patient’s cancer responds to therapy and whether it has recurred after therapy. The particles are currently being developed further by the researchers with the intention of testing them on humans. Glympse Bio, an organization helped to establish by Bhatia, has performed stage 1 clinical preliminaries of a previous form of the urinary symptomatic particles and viewed them as protected in patients.
More information: Liangliang Hao et al, CRISPR-Cas-amplified urinary biomarkers for multiplexed and portable cancer diagnostics, Nature Nanotechnology (2023). DOI: 10.1038/s41565-023-01372-9