A further improvement in nuclear power microscopy currently makes it conceivable to simultaneously picture the level profile of nanometer-fine designs as well as the electric flow and the frictional power at strong fluid connection points. A group from the Helmholtz-Zentrum Berlin (HZB) and the Fritz Haber Establishment (FHI) of the Maximum Planck Society has prevailed with regards to examining electrocatalytically dynamic materials and acquiring experiences that will assist with streamlining impetuses. The technique is additionally possibly reasonable for concentrating on processes on battery cathodes, in photocatalysis, or on dynamic biomaterials.
To deal with the energy change, it will likewise be critical to quickly foster modest and effective materials that can be utilized to separate water or CO2 by electrocatalysis. In this cycle, some portion of the electrical energy is stored in the synthetic response items. The proficiency of such electrocatalysts relies generally upon the idea of the cathode-electrolyte interfaces, for example, the points of interaction between the strong cathodes and the commonly watery electrolyte. Nevertheless, spatially settled actual investigations of such strong fluid points of interaction are still moderately scant.
More experiences with AFM
Dr. Christopher S. Kley and his group have now fostered another way to deal with complementary nuclear power microscopy (AFM). A very sharp tip is examined across the surface, and its level profile is recorded. By joining the tip to the furthest limit of a scaled-down cantilever, the power connections between the tip and the example surface, including frictional powers, can be estimated with high responsiveness.
“We were able to clearly detect islands of copper oxide with higher electrical resistance, as well as grain boundaries and low-conductivity regions in the hydration layer where the catalyst surface comes into contact with the aqueous electrolyte,”
Dr. Martin Munz, first author of the study.
What’s more, the electrical flow moving through the mechanical contact can be estimated when a voltage is applied. “This permitted us to all the while decide the electrical conductivity, the mechanical-substance grating, and the morphological properties in situ (for example, under the applicable fluid stage conditions as opposed to in vacuum or in air),” says Kley.
Copper-gold electrocatalyst
Utilizing this technique, the researchers are currently contemplating a nanostructured, bimetallic copper-gold electrocatalyst in a joint effort with Prof. Beatriz Roldán Cuenya from the Fritz-Haber-Foundation (FHI). Among others, such materials are utilized in the electrocatalytic change of CO2 into energy transporters.
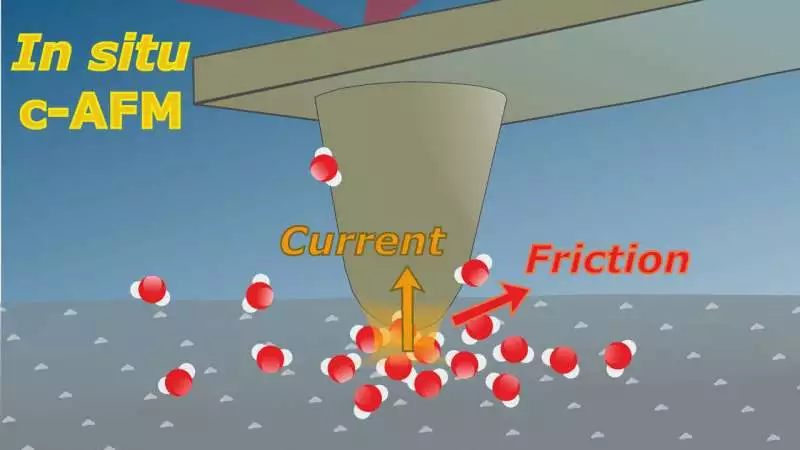
The guideline of corresponding nuclear power microscopy in contact mode is: a fine tip toward the finish of a cantilever filters the surface. This permits force associations between the tip and the example surface to be estimated, including frictional powers. On the off chance that a voltage is also applied, the electric flow moving through the contact can likewise be estimated.
“We had the option to clearly distinguish islands of copper oxide with higher electrical opposition, yet additionally grain limits and low-conductivity areas in the hydration layer where the impetus surface comes into contact with the watery electrolyte,” says Dr. Martin Munz, the first creator of the review.
Such outcomes at impetus electrolyte interfaces help to streamline them in a designated way. “We can now see how nearby electrochemical conditions impact charge movement at the point of interaction,” says Kley.
“Nevertheless, our outcomes are likewise of general interest to energy research, particularly for the investigation of electrochemical change processes, which likewise assume a part in battery frameworks.”
Bits of knowledge about strong fluid connection points can likewise be valuable in totally various areas of exploration, for example, understanding erosion processes, nanosensor frameworks, and perhaps tending to logical questions in fluidics and ecological sciences, for example, disintegration or statement processes on metal surfaces presented to water.
The discoveries are distributed in the Diary of the American Compound Society.
More information: Martin Munz et al, Nanoscale Electron Transfer Variations at Electrocatalyst–Electrolyte Interfaces Resolved by in Situ Conductive Atomic Force Microscopy, Journal of the American Chemical Society (2023). DOI: 10.1021/jacs.2c12617