Scalable methods to reliably split water into hydrogen and oxygen could have important repercussions for the energy sector, as hydrogen inside fuel cells can generate electrical power. These techniques may contribute to the reduction of Earth’s emissions of greenhouse gases by producing a lot of hydrogen for more environmentally friendly energy sources.
Photocatalysts, which are substances that are able to absorb light and use the energy of that light to start chemical reactions, are one method for splitting water molecules into hydrogen and oxygen. This method basically involves shining light on these materials, which starts the reaction that turns water molecules into hydrogen and oxygen.
New hybrid photocatalysts with an internal quantum efficiency above 100 percent were recently developed by researchers at Northwestern Polytechnic University in China. These materials, which were presented in a Nature Energy paper, were found to overcome some of the drawbacks of photocatalytic systems that had been proposed previously for water splitting processes.
“Researchers have made multiple attempts over the past ten years to reach a solar-to-hydrogen efficiency of more than 10%, which is a competitive benchmark efficiency in the market for hydrogen.”
Dr. Xuanhua Li, one of the researchers who carried out the study,
Dr. Xuanhua Li, one of the study’s authors, told Tech Xplore, “Researchers have made numerous attempts over the past decade to achieve a solar-to-hydrogen efficiency of more than 10%, which is a competitive benchmark efficiency in the hydrogen market.”
“Over a wide range of excitation wavelengths, the photocatalyst’s internal quantum efficiency (the ratio of the number of incident photons absorbed to twice the amount of hydrogen produced) must reach a moderately high value (ideally >100%) to achieve this goal.”
Since the majority of previously reported efficiencies were insufficient to enable the widespread use of water splitting processes, a number of previous studies attempted to develop useful strategies for increasing the quantum efficiency of photocatalysts and photoelectric devices. The so-called multiple exciton generation (MEG) effect, in which a nanocrystal quantum dot absorbs a single photon to generate multiple excitons, was found to be a particularly promising strategy.
Dr. Li elaborated, “For instance, one study demonstrated that the quantum efficiency of lead-salt nanocrystals increases roughly linearly with pump-photon energy and demonstrates a maximum quantum efficiency of up to 700 percent.” In photoelectro chemical cells for the generation of hydrogen, IQEs exceeding 100 percent can be achieved by layer-by-layer depositing of PbS quantum dots on top of fluorine-doped tin oxide (TiO2). However, due to the addition process for electric energy to hydrogen energy, demonstrations of the MEG effect in particulate photocatalytic water splitting systems are still rare in comparison to photoelectric devices.
New MEG-effect photocatalysts for efficient water splitting were the primary focus of recent research by Dr. Li and his colleagues. They believed that these materials would be a viable option for the scalable production of hydrogen if their internal quantum efficiency exceeded 100 percent.
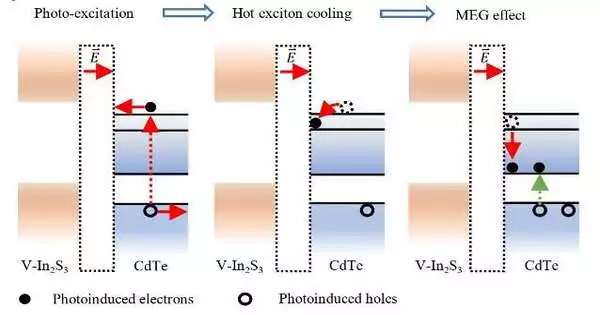
The researchers needed to create an interfacial trapping state and a powerful built-in interfacial electric field in order to construct these effective photocatalysts. The multiple exciton generation (MEG) effect in photocatalytic water splitting would then be sufficiently motivated by this.
“We created mixture photocatalysts involving CdTe quantum dabs and V-doped In2S3 (CdTe/V-In2S3),” Dr. Li said. “Specific to the CdTe/V-In2S3 interface, an increase in the Fermi level difference and consequently a 14.14 fold increase in the built-in electric field intensity can be observed when both the quantum dot size and the quantity of V-dopants are increased. This is because the Fermi levels of the CdTe quantum dots shift downward and those of V-In2S3 shift upward. At the CdTe/V-In2S3 interface, an interfacial state containing In 5s and S 3p orbitals was generated in the meantime.”
A hot electron and a hole are produced when a CdTe quantum dot in the team’s photocatalyst is excited during the photocatalytic process. Driven by the materials’ implicit electric field, the hot electron in the conduction band of the CdTe quantum dab is moved from CdTe to V-In2S3 and eventually caught at the CdTe/V-In2S3 interface in an interfacial state comprised of In 5s and S 3p orbitals.
According to Dr. Li, “the strong built-in electric field and interfacial state at the CdTe/V-In2S3 interface slow the relaxation rate of the hot electrons, enabling hot electrons with sufficient excess energy to undergo MEG” in contrast to conventional photocatalysts. At an excitation wavelength of 350 nm, the photocatalyst achieves an internal quantum efficiency of around 114 percent, which is, to our knowledge, the highest value among all reported photocatalysts for overall water splitting. The effective utilization of MEG in photocatalytic water splitting is made possible by our optimization of the interfacial built-in electric field and interfacial state.”
The hybrid photocatalysts developed by this group of researchers showed higher internal quantum efficiencies than any other photocatalyst that had been proposed previously for water splitting, which was very encouraging in the initial evaluations. Later on, this new work could prepare for the huge scope of photocatalytic water parting.
Photocatalytic devices that operate in the MEG regime can now be designed in new ways thanks to the design strategy that Dr. Li and his colleagues presented. This could before long prompt the advancement of extra materials and arrangements with progressively high quantum and sunlight-based hydrogen efficiencies, which could additionally advance the utilization of sun-powered energy to create hydrogen.
Dr. Li continued, “It should be noted that the competition for light absorption between V-In2S3 and the CdTe quantum dots may limit the photocatalytic overall water splitting ability of CdTe/V-In2S3.” Besides, accomplishing high quantum productivity over many frequencies is vital to advancing the common sense of this innovation. To further develop this examination, we expect to foster more effective photocatalysts with bigger underlying electric field forces and various valence band interfacial states, for example, by building a Janus structure.”
Dr. Li and his colleagues also intend to develop new non-MEG/MEG heterojunctions with a wide absorption range in their subsequent research. By joining frequency-corresponding MEG parts with non-MEG parts, they desire to additionally work on the photocatalysts’ general execution.
More information: Youzi Zhang et al, Internal quantum efficiency higher than 100% achieved by combining doping and quantum effects for photocatalytic overall water splitting, Nature Energy (2023). DOI: 10.1038/s41560-023-01242-7