Researchers at VCU Massey Disease Center have uncovered a formerly unnoticed connection between two arrangements of proteins that work together to fight off the development of pancreatic malignant growth. The discoveries could provide significant data for the improvement of novel treatments for what is right now a hopeless sickness.
Pancreatic ductal adenocarcinoma (PDAC) represents by far most of every pancreatic growth and is the fourth-leading reason for disease-related deaths around the world. Most patients are analyzed at a high level when the infection is inoperable and there are no powerful treatments.
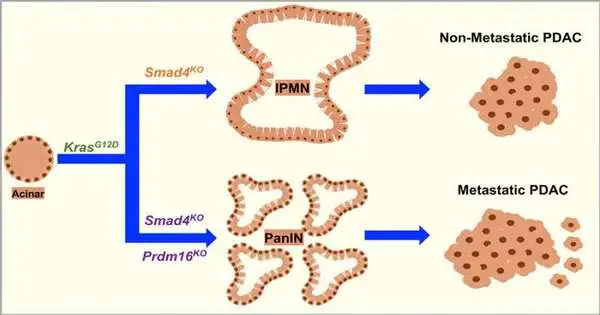
New exploration is distributed in the Diary of Cell Science, driven by Azeddine Atfi, Ph.D., head of the Disease Science research program who holds the Mary Anderson Harrison Recognized Residency in Malignant Growth. Exploration at Massey found that the concurrent enactment of two quality-directing proteins — Prdm16 and Smad4 — is related to a reduction in pancreatic disease movement.
“Our findings shed light on a previously unknown interaction between Smad4 and Prdm16, which appears to influence the growth trajectory of pancreatic cancer and could eventually pave the way for innovative therapeutic solutions to combat this devastating illness,”
Atfi, who is also a professor in the Department of Biochemistry and Molecular Biology.
“Our review reveals insight into a formerly uncharacterized transaction somewhere in the range of Smad4 and Prdm16, which seem to direct the development direction of pancreatic malignant growth, that could eventually make ready for creative, helpful forward leaps to control this dangerous sickness,” said Atfi, who is likewise a teacher in the Branch of Natural Chemistry and Sub-atomic Science at the VCU Institute of Medication.
The shortfall of Smad4 has recently been associated with disease development in PDAC cells. Prdm16 controls different fundamental cell processes, including the development of tissues and organs. The protein was first noticed experimentally in leukemia, where past examination demonstrated that it could work as a growth silencer. It had not, however, been concentrated on in pancreatic disease.
Atfi and his exploration group found that when both Prdm16 and Smad4 were inactivated, the pancreatic growths developed forcefully and framed metastatic sores in the lung. Notwithstanding, they likewise resolved that when Smad4 was inactivated alone, Prdm16 became overexpressed and contributed to disease development.
At the point when the examination group noticed cells with the two proteins enacted — where Prdm16 was found downstream along a similar hereditary informing pathway as Smad4 — Prdm16 effectively stifled growth movement and metastasis in pancreatic disease cells, recommending the two of them play a significant, consolidated role in fending off disease cells.
“Consider Smad4 a film chief advising Prdm16 how to satisfy the job of a malignant growth warrior. Without Smad4 as the chief, Prdm16 begins to fill the role of a malignant growth ally,” Atfi said. “Our discoveries highlight a formerly unknown system that coordinates the antitumor job of Prdm16 and further shed new experiences into the sub-atomic reason for pancreatic disease.”
More exploration should be led to solidly lay out whether Prdm16 inspires anti-growth action at the beginning phases, assuming this happens through coordinated impacts on disease cell development or through the reinventing of the cancer microenvironment. Atfi said a more extensive examination of how Smad4 and Prdm16 interface could assist with distinguishing extra hereditary players that could be designated through disease treatment
“We guess that our revelation will direct approaching examinations trying to comprehend and disentangle the cell standards of pancreatic disease,” Atfi said.
More information: Eric Hurwitz et al, Antagonism between Prdm16 and Smad4 specifies the trajectory and progression of pancreatic cancer, Journal of Cell Biology (2023). DOI: 10.1083/jcb.202203036