Using an advanced nuclear approach, ANSTO discovered the first experimental evidence to support a newly published universal law that provides insights into the complex energy states of liquids.
The study was just published as the editor’s choice in the Publication of Physical Chemistry Letters, and it was highlighted on the front cover of the journal.
Alessio Zaccone and Matteo Bagglioli published their equation for the vibrational density of states in PNAS in 2021, offering an answer to a question that had eluded scientists for at least a century.
The difficulty of determining the distribution of these complex energy states for liquids has been answered by an elegant mathematical theory.
The distribution of the frequencies or vibrational energy of the waves that propagate in the material is one of the most important parameters in matter physics. It’s crucial since it’s the beginning point for calculating and comprehending some fundamental properties of matter, like specific heat and thermal conductivity, as well as the light-matter interaction, “Prof. Zaccone stated on the website of the University of Milan.
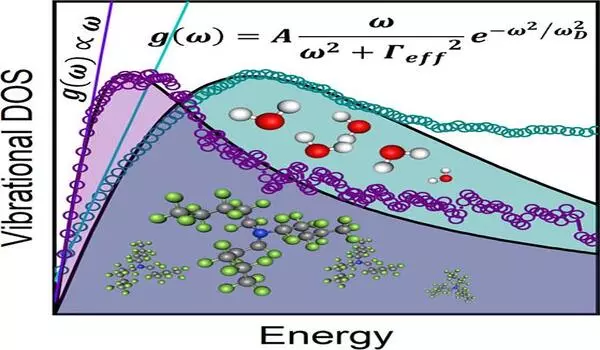
The major issue with liquids is that, in addition to acoustic waves, there are other types of vibrational excitations related to low energies of disordered motion of atoms and molecules that are almost non-existent in solids. These excitations are often short-lived and linked to the dynamic chaos of molecular motions, but they are still abundant and essential, particularly at low energies. These excitations, known in the specialized literature as “instantaneous normal modes” or INMs, are difficult to deal with mathematically since they correspond to energy states defined by imaginary numbers.
The vibrational densities of states for numerous liquid systems, including water, liquid metal, and polymer liquids, have been measured using the time-of-flight neutron spectrometer Pelican at ANSTO’s Center for Neutron Scattering. The Pelican instrument can measure rotational and translational vibrations over short time intervals and at low energy because of its great sensitivity.
The investigations at ANSTO validated Alessio Zaccone and Matteo Bagglioli’s prediction of a linear connection between the vibrational density of states and frequency at low energies, as illustrated in the figure below.
With no access to instruments due to the COVID lockdown, the small team, which included University of Wollongong Ph.D. candidate Caleb Stamper, Dr. Cortie, and Dr. Yu, decided to focus on re-analyzing past experimental data from a new perspective in order to validate the new law, which was inspired by Alessio Zaccone and Matteo Bagglioli’s theoretical work.
“The exercise not only achieves such a great outcome but also provides a good introduction of neutron spectroscopy to Caleb, who has done an excellent job,”
Dr. Yu as Caleb’s ANSTO
Caleb’s ANSTO supervisor and the corresponding author of the work, Dr. Yu, commented, “The exercise not only achieves such a remarkable conclusion but also provides a good introduction to neutron spectroscopy for Caleb, who has done an excellent job.”
Their work on thermoelectric materials would also aid them in addressing problems related to phase transitions in superionic liquids.
Liquids are not mechanically stable, since the atoms in a liquid diffuse and the liquid as a whole will flow.”
The universal rule is based on a theoretical framework called instantaneous normal modes, which prescribes a set of instantaneous forces, frequencies, and velocities as quantities, as defined by Prof. Zaccone above.
The presence of a small proportion of “imaginary modes” caused a challenge in developing a theory to predict the vibrational density of states in liquids.

“Imaginary modes are significant because they depict the fact that a liquid is unstable.” A liquid’s atoms constantly interact with one another, but not in the same manner that a solid’s do. The relationship is not “harmonic,” which means that following an encounter, the atoms will not be returned to their original form. “The atoms will continue to diffuse rapidly and pass each other,” Stamper predicted.
“The imaginary modes reflect a liquid’s potential energy surface’s negative curvature.” It’s a complicated energy landscape, but think of it like a surfer riding a wave in the ocean. The wave’s curves are followed by the atoms in the liquid (see the front cover of the journal). However, the atoms can be on the crest, under the surfboard, or in the trough, always moving, “Dr. Yu explained.
“The law will be just as important for liquids as the Debye law is for solids. It will serve as the cornerstone for the entire area of liquid research and beyond. “





