Fluid energizes with high energy thickness are essential in many applications where compound energy is converted into controlled movement, such as rockets, gas turbines, boilers, and certain vehicle motors.Other than their burning qualities and execution, it means a lot to ensure the wellbeing and security of these fuels when being used, as well as during transport and capacity.
One normal danger while managing fluid energies is that they can dissipate rapidly whenever given space, creating billows of exceptionally combustible gases. As one would expect, this can prompt horrendous blasts or fire mishaps. To handle this issue, specialists have thought about the utilization of gelled fills, or energizes transformed into thick gel-like substances by cold temperatures. In any case, there are numerous viewpoints to upgrade and obstacles to defeat before gelled fills can go past the exploration stage.
“Polymeric gel storage could avoid explosions and fires by dramatically lowering fuel evaporation and, as a result, the creation of combustible gaseous mixtures, which can easily occur following a leak in a storage facility. Much work remains to be done on this front, including testing the stability and performance of polymeric gels at various temperature, pressure, and humidity settings, as well as developing easier manufacturing techniques and better ways to use these fuel-loaded gels in real engines.”
Prof. Hosoya explains
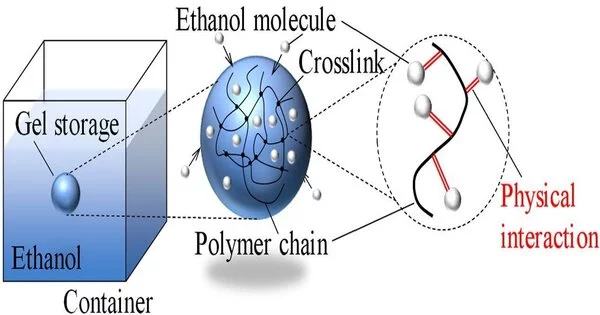
A group of analysts led by Prof. Naoki Hosoya from Shibaura Institute of Technology (SIT) and Prof. Shingo Maeda from Tokyo Institute of Technology (Tokyo Tech), Japan, recently explored a really convincing answer to the security issue of fluid powers, to be specific, putting them away inside polymeric gel organizations. In their review, the group investigated the exhibit, benefits, and constraints of putting away ethanol, a typical fluid fuel, inside an artificially cross-connected poly (N-isopropylacrylamide) (PNIPPAm) gel. This paper was published in the Chemical Engineering Journal.
In the first place, they checked whether catching ethanol atoms inside the long and artificially entwined PNIPAAm polymer chains decreased their dissipation rate. To test this, the scientists made little circles of PNIPAAm gel stacked with ethanol and put them on an electronic scale to record how mass changed as ethanol disintegrated. They likewise played out this trial with a comparable puddle of ethanol, with generally a similar surface region and mass as the gel circle.
They found that putting away ethanol inside the polymer gel totally stifled the fuel’s propensity to disintegrate quickly. This is probable because of how ethanol atoms are “caught” in the gel, as Prof. Hosoya makes sense of that “the polymeric gel contains incalculable three-layered polymer chains that are artificially cross-connected in major areas of strength for a These chains tie the ethanol particles through different actual collaborations, restricting their vanishing all the while. Interestingly, the stacked gel doesn’t act like a wet towel. Though a wet towel would deliver its fluid whenever wrung, the polymeric gel didn’t let out ethanol effectively under outer powers.
With the issue of dissipation addressed, the group continued on to inspect the real ignition attributes of the ethanol in the polymeric gel organization to check whether they were consumed productively. They lighted ethanol-stacked gel circles of different sizes and noticed the progressions in their mass and shape profiles progressively. In light of this, they established that the consuming of the stacked PNIPAAm gel circles was comprised of two stages: a stage overwhelmed by unadulterated ethanol consuming, followed by a subsequent stage overwhelmed by the consuming of the PNIPAAm polymer itself.
Through an ensuing hypothetical examination of these outcomes, the group reached a significant resolution: the first and primary ignition period of the stacked PNIPAAm gel circles follows a consistent drop temperature model, otherwise called the “d2 regulation.” What this implies is that the consumption of the ethanol-stacked gel can be depicted by a similar model utilized for fluid fuel drops, indicating that their burning exhibitions ought to be comparable.
Generally, this study is a stepping stone towards better approaches to securely moving and storing fluid energy inside polymer gels, which could save many lives. “Polymeric gel stockpiling could forestall blasts and fire mishaps by radically lessening the vanishing of fills and, thusly, the development of combustible vaporous blends, which can promptly happen following a hole in a storage space,” makes sense to Prof. Hosoya. Much work actually still needs to be done on this front. For example, actually taking a look at the solidness and execution of polymeric gels at various temperature, strain, and moisture conditions, as well as creating a less difficult manufacturing methodology and better ways of utilizing these fuel-stacked gels in genuine motors.”