A substance that destroys a protein that drives cancer, including B-cell lymphoma, has been developed by researchers at the Institute of Cancer Research in London.
The “molecular glue” degrader will be a powerful tool for studying cancer biology and could eventually be developed into a cancer drug with additional research.
It is an illustration of a novel approach to drug discovery known as protein degradation, which aims to remove proteins that can cause disease from the cell’s natural disposal system in order to target cancer.
“One intriguing aspect of our BCL6 project was that the first compound discovered through our screen had low solubility, which is not ideal for drugs.”
Dr. Benjamin Bellenie, senior staff scientist in the ICR’s Centre for Cancer Drug Discovery,
The researchers describe in a new study how the degrader selectively binds to the BCL6 protein, which B-cell lymphoma cells require for survival, and tags it for destruction.
BCL6 regulates genes involved in cell division and death by binding to DNA. Many cancers are caused by mutations in the protein, so reducing or inhibiting its levels could slow their growth.
An inventive revelation
The analysts looked to find intensities that disturb the capability of the BCL6 protein and repress lymphoma cell development. Their previous work prompted the identification of a few mixtures that could be grown further as BCL6 inhibitors.
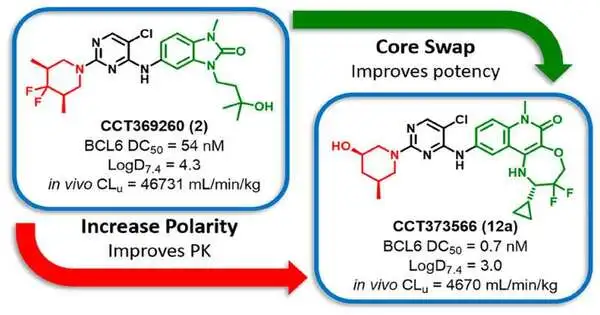
The group saw that a portion of the inhibitors they made likewise had the additional capacity to debase the BCL6 protein. Although these small-molecule degraders were able to completely deplete BCL6 in cells, they were difficult to dose in sufficient quantities in animal models to maintain BCL6 levels due to their low solubility in water.
The team presents the first BCL6 molecular glue-type degrader with suitable properties for testing in animal models in their most recent paper, which was published in the Journal of Medicinal Chemistry. This paper builds on the team’s previous findings.
The team was able to create a potent compound that can bind to BCL6 more tightly and has improved water solubility, enabling sustained degradation of the protein in tumors in mice. They did this by understanding what features of the molecule were required to give the degrader effect.
In studies conducted on mice, the new compound had a negligible effect on the levels of BCL6, with tumors receiving the drug growing significantly more slowly than control tumors that did not receive any treatment.
Although the initial results in mice were insignificant, they do suggest that, when used in conjunction with other treatments, BCL6-targeted molecular glue-type degraders might be able to control lymphoma tumor growth.
The medication’s capacity to support the consumption of BCL6 likewise makes it an exceptionally useful asset to concentrate on BCL6 science.
How do degraders resembling molecular glue function?
Cells go through a fundamental process called “protein turnover.” It is constrained by the ubiquitin proteasome framework, where specific chemicals, including a group of proteins called the E3 ubiquitin ligases, perceive inadequate proteins and label them with a little administrative protein called ubiquitin.
A different part of the system finds the ubiquitin-tagged protein and takes it to the proteasome, a protein complex that breaks down damaged or unnecessary proteins.
Small compounds known as molecular glue-type degraders make it easier for a target protein to interact with the ubiquitin proteasome system. They act in a similar way to more customary ‘little particle’ drugs, which tie to a particular site on a protein and block its capability.
Even though many small-molecule drugs have been very good at treating cancer, the protein degradation method gives hope for new therapies that can hit targets that are “undruggable” right now and might work for a long time by removing all proteins from cells.
“One interesting feature of our BCL6 project was that the initial compound we discovered from our screen had low solubility, which is not an ideal feature for drugs,” stated Dr. Benjamin Bellenie, co-leader of the Complex Chemistry Study and senior staff scientist in the ICR’s Center for Cancer Drug Discovery.
“With additional examinations, we understood that the part adding to low solvency was additionally giving the compound its ‘stick-like’ trademark. This made it extremely testing to streamline the particle, as any progressions to further develop its solvency frequently brought about loss of its capacity to successfully adhere to the objective.”
“As a result, we are overjoyed to announce that we have discovered a new set of molecular glue-type degraders for BCL6, the protein that drives lymphomas and other cancers.” “Our undertaking was a sensitive equilibrium of enhancing dissolvability of our up-and-comer compounds without losing power, and at last, we had the option to make a compound that can tie BCL6 firmly and structure a “stuck complex” even at low focuses.”
Concentrate on co-pioneers. Teacher Swen Hoelder, head of science at the ICR, said, “We’re extremely energized by the capability of protein debasement to convey new medications that could have a major effect in malignant growth treatment, and that is the reason we established our Middle for Protein Debasement here at the ICR. I am pleased to announce the discovery of our new BCL6 molecular glue-type degrader in this new study, which is also an excellent example of creative science in a cutting-edge cancer drug discovery field.”
More information: Rosemary Huckvale et al, Improved Binding Affinity and Pharmacokinetics Enable Sustained Degradation of BCL6 In Vivo, Journal of Medicinal Chemistry (2022). DOI: 10.1021/acs.jmedchem.1c02175