Utilizing the CRISPR-Cas9 quality altering framework, UT Southwestern scientists revised the changes liable for a typical acquired heart condition called widened cardiomyopathy (DCM) in human cells and a mouse model of the illness. Their discoveries, distributed in Science Translational Medication, may one day give hope to an expected 1 out of every 250 individuals overall who experience the ill effects of this condition.
“All of the qualities of illness we see due to these changes were switched with CRISPR-Cas9 treatment. “Most would agree the outcome of this approach totally surpassed our assumptions,” said Eric Olson, Ph.D., Seat and Teacher of Sub-atomic Science at UTSW, who co-led the review with partners Rhonda Bassel-Duby, Ph.D., Teacher of Atomic Science, and Takahiko Nishiyama, M.D., Ph.D., a postdoctoral individual in the Olson lab.
DCM is brought about by changes in a quality known as RNA-restricting motif protein 20 (RBM20), which influences the creation of many proteins in cardiovascular muscle cells liable for the heart’s siphoning activity. This illness unleashes broad ruin all through the heart, slowly annihilating its capacity to agree and making it become very extended and explosive after some time. Treatment is restricted to drugs, which can work on contractile capability but don’t give a long-lasting fix, or a heart transplant, which regularly isn’t a choice because of a lack of benefactor organs.
“CRISPR-Cas9 therapy was able to completely reverse all of the disease characteristics we see as a result of these mutations. It’s fair to say that the effectiveness of this strategy far surpassed our expectations.”
Eric Olson, Ph.D., Chair and Professor of Molecular Biology at UTSW
Drs. Olson, Bassel-Duby, Nishiyama, and their colleagues focused on CRISPR-Cas9, a well-known device for hereditary exploration that was awarded the Nobel Prize in Science in 2020.Utilizing this framework, analysts might possibly address illnesses causing changes in significant qualities.
So far, the Food and Medication Organization has endorsed a single clinical study that utilizes this innovation to attempt to treat sickle cell illness. In any case, Dr. Olson said CRISPR-Cas9 can possibly treat an untold number of other hereditary illnesses. Dr. Olson and colleagues used CRISPR-quality altering to create a method to stop the movement of Duchenne muscular dystrophy in animal models.
To test the efficacy of this method for treating DCM, the researchers used an infection to deliver CRISPR-Cas9 components to heart muscle cells derived from human cells that had undergone two distinct types of DCM-causing changes.Researchers utilized this quality-altering innovation to trade a single nucleotide, the essential unit of DNA, to address one kind of change. In another cell arrangement, scientists replaced a piece of DNA from altered RBM20 with a sound portion of this quality.
After CRISPR-Cas9 treatment, the freak cells gradually lost DCM-inherent properties: the protein produced by RBM20 moved to its expected location in the core, and the cells began producing solid proteins.
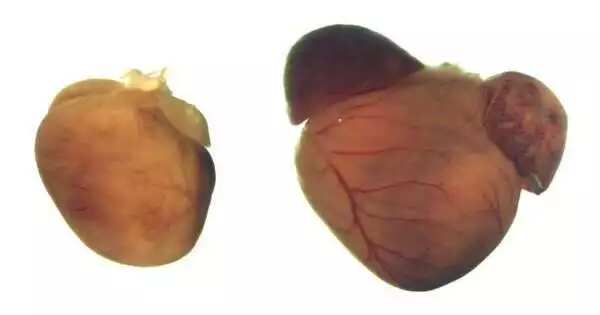
When the researchers administered the CRISPR-Cas9 treatment to 1-week-old mice, the creatures never developed expanded hearts and had normal life expectancies.Untreated mice had side effects resembling those of human DCM patients.
According to the researchers, a few challenges must be overcome before this treatment can be used in DCM patients.Work is required to ensure that the effects of CRISPR-Cas9 are long-lasting and precise, and that the smallest amount possible is conveyed.Similarly, it is unclear whether the treatment could be used in patients whose illnesses have progressed.In any case, Dr. Olson said he’s hopeful that this framework could be utilized to treat various other familial illnesses.
“The speed of this field is truly amazing,” he said. “That’s what I expect if this moves forward into patients; we’re not talking in many years—we’re talking in no time.”
More information: Takahiko Nishiyama et al, Precise genomic editing of pathogenic mutations in RBM20 rescues dilated cardiomyopathy, Science Translational Medicine (2022). DOI: 10.1126/scitranslmed.ade1633