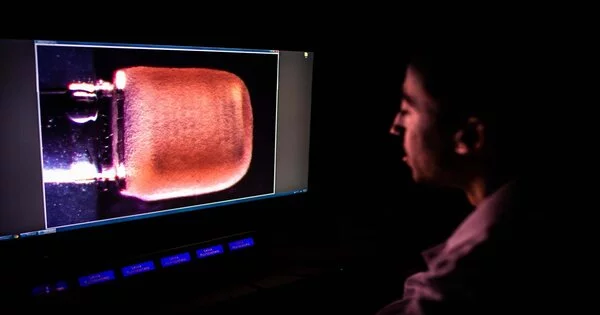
There’s no safe method to gain a close-up view of the human heart at work: you can’t just take it out, look at it, and put it back in. Scientists have tried a variety of approaches to get past this fundamental obstacle, including attaching lab-grown heart tissues to springs to watch them expand and contract, and hooking up cadaver hearts to machinery to encourage them to pump again. Each method has drawbacks: reanimated hearts can only beat for a few hours, and springs can’t simulate the forces at work on a real muscle. However, gaining a deeper understanding of this crucial organ is critical because, according to the Centers for Disease Control and Prevention, someone in America dies of heart disease every 36 seconds.
Now, an interdisciplinary group of engineers, biologists, and geneticists has created a small duplicate of a heart chamber using nanoengineered parts and human heart tissue. There are no springs or external power sources—it simply beats on its own, powered by live heart tissue generated from stem cells, just like the genuine thing. Researchers may use the gadget to gain a better understanding of how the organ operates, allowing them to follow how the heart develops in the embryo, analyze the impact of disease, and test the efficacy and side effects of new treatments—all without putting patients at risk and without leaving the lab.
“Heart disease is the number one cause of death in the United States, touching all of us. Today, there is no cure for a heart attack. The vision of CELL-MET is to change this.”
says White, who was chief scientist at Alcatel-Lucent Bell Labs before joining BU in 2013.
The team behind the device, dubbed miniPUMP and officially known as the cardiac downsized Precision-enabled Unidirectional Microfluidic Pump, claims that the invention could pave the way for lab-based replicas of other organs, such as the lungs and kidneys. Their findings were reported in the journal Science Advances.
“We can investigate disease progression in ways that have never been feasible before,” explains Alice White, chair of mechanical engineering at BU College of Engineering. “We opted to concentrate on heart tissue because of its complex mechanics, but we demonstrated that when nanotechnology is combined with tissue engineering, it is possible to replicate this for multiple organs.”
According to the researchers, the device could someday speed up and reduce the cost of drug development. Rather than investing millions of dollars (and possibly decades) in moving a therapeutic medication through the development pipeline only to have it fail at the final hurdle when tested in humans, researchers might utilize the miniPUMP from the start to better predict success or failure.
Cell-MET is a multi-institutional National Science Foundation Engineering Research Center in Cellular Metamaterials led by BU. The center’s mission is to regenerate injured human heart tissue by bringing together scientists and industry experts to test new medications and construct artificial implanted patches for hearts damaged by heart attacks or illness.
Individualized medicine
There are numerous things that can go wrong with your heart. The heart’s two top and two bottom chambers keep your blood moving so that oxygen-rich blood flows and feeds your body when all four cylinders are functioning properly. When disease occurs, however, the arteries that carry blood away from your heart might narrow or get blocked, valves can leak or malfunction, the heart muscle can thin or thicken, or electrical signals can shorten, resulting in too many—or too few—beats. Heart disease, if left untreated, can cause discomfort (such as shortness of breath, exhaustion, swelling, and chest pain) and, in many cases, death.
As it pumps blood through our bodies, the heart is subjected to complicated stresses, explains Christopher Chen, the William F. Warren Distinguished Professor of Biomedical Engineering at BU. And, while we know that aberrant forces, such as high blood pressure or valve disease, cause the heart muscle to alter for the worse, it has been difficult to duplicate and analyze these disease processes. That’s why we sought to create a little heart chamber. “
The miniPUMP is about the size of a postage stamp, measuring 3 square centimeters. Its custom-made components are put onto a thin sheet of 3-D-printed plastic and are designed to function like a human cardiac ventricle (or muscular lower chamber). There are miniature acrylic valves that open and close to control the flow of liquid—in this case, water rather than blood—and little tubes that funnel that fluid in the same way as arteries and veins do. And thumping away in one corner are cardiomyocytes, stem cell-derived muscle cells that contract heart tissue.
They’re made from induced pluripotent stem cells, “explains Christos Michas (ENG ’21), a postdoctoral researcher who devised and led the creation of the miniPUMP as part of his Ph.D. thesis.
To create the cardiomyocyte, researchers take an adult cell—it might be a skin cell, a blood cell, or almost any other cell—reprogram it into an embryonic-like stem cell, and then transform it into the heart cell. The cardiomyocytes not only give the device a real heart, but Michas believes they also provide the system with immense promise in helping to pioneer tailored medications. Researchers may insert a damaged tissue into the gadget, then test a medicine on that tissue to see how its pumping ability is affected.
“With this technique, I can observe how the medicine will react in you because these are your cells,” Michas explains. This technology better replicates certain of the heart’s functions while also allowing us to have different people that it replicates. It’s a more accurate model for predicting what would happen in humans without really going into humans. “
According to Michas, this could allow scientists to assess the possibility of a novel heart disease drug’s success long before it enters clinical trials. Many medication candidates fail due to negative side effects.
At the start, when we’re still experimenting with cells, we can incorporate these devices and have more precise forecasts of what will happen in clinical trials, Michas says. “It also implies that the medications may have fewer adverse effects.”
A hair is thinner than a human hair.
An acrylic scaffold that supports and travels with the heart tissue as it contracts is a critical component of the miniPUMP. The scaffold resembles an artistic piston, with its sequence of superfine concentric spirals—thinner than a human hair—connected by horizontal rings. It’s an important element in the puzzle, providing structure to the heart cells (which would otherwise be formless blobs) while exerting no active force on them.
“We don’t think past ways of analyzing cardiac tissue represent the way the muscle would behave in your body,” says Chen, who is also the director of BU’s Biological Design Center and an associate faculty member at Harvard’s Wyss Institute for Biologically Inspired Engineering. “This is our first chance to develop something mechanically sound.” This provides us with the first opportunity to design something mechanically akin to what we believe the heart is experiencing—a major step forward.
The scientists used two-photon direct laser writing, a more precise kind of 3-D printing, to print each of the small components. When light is shone into a liquid resin, the places it touches solidify. Since the light can be aimed with such precision—focused on a tiny spot—many of the miniPUMP’s components are measured in microns, which is smaller than a dust particle.
“The structural elements are so delicate that things that would normally be stiff are flexible,” White adds. “Think of optical fiber as an analogy: a glass window is incredibly inflexible, yet you can wrap a glass optical fiber around your finger.” Acrylic can be very strong, but at the scale involved in the miniPUMP, the beating cardiomyocytes can compress the acrylic scaffold.”
According to Chen, the pump’s scale demonstrates “that with finer printing designs, you might be able to generate more sophisticated cell organizations than we thought was conceivable before.” Currently, when researchers try to make cells, whether heart cells or liver cells, he adds, they’re all disorganized—”to acquire structure, you have to cross your fingers and hope the cells create anything.” This indicates that the tissue scaffolding pioneered in the miniPUMP has far-reaching potential beyond the heart, setting the groundwork for other organs-on-a-chip ranging from kidneys to lungs.