The quality altering procedure known as CRISPR has sped up organic and clinical exploration somewhat recently by permitting researchers to fix the DNA of human cells nearly as simply as utilizing some scissors.
Quality altering — particularly CRISPR, because it is simple to use — has given researchers hope for the restoration of hereditary infections, including disease.
Presently, a group of scientists at Case Western Reserve University has fostered a strategy that could make CRISPR techniques considerably more precise—and really encouraging.
“This new technology allows us to target not only the location, but also the time of the alteration—and to halt it. That has never been done before, and we believe it might be very significant.”
Fu-Sen Liang, associate professor of chemistry,
Their new method, detailed in a recent focus published in the journal Nature Communications, combines CRISPR with a synthetic interaction that allows quality altering to be more definitively found and coordinated.
Such accuracy makes the innovation more powerful and lessens expected secondary effects, said Fu-Sen Liang, academic partner of science, who led the exploration group.
While the discoveries are still in their early stages, the scientists, who include a few Case Western Reserve post-doctoral analysts and understudies, believe the work could lead to more effective treatment for certain infections, including cancer.
“What we are doing is investigating one more method for changing the result of the quality by focusing on the RNA with a compound,” Liang said. “This new innovation empowers us to target the spot, yet the hour of the adjustment — and the capacity to stop it.” That has never been conceivable, and we accept it very well may be vital. “
The procedure
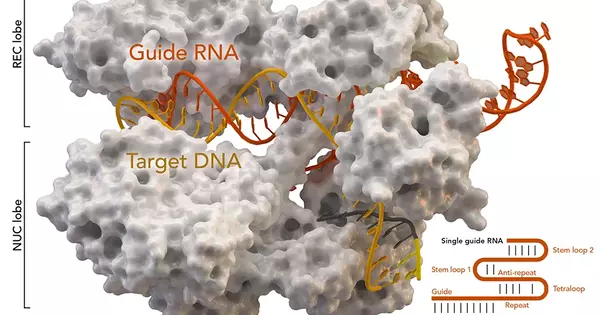
CRISPR is an abbreviation for Clustered Regularly Interspaced Short Palindromic Repeats. The remarkable innovation, revealed in 2012, employs a specific catalyst—directed by RNA, or ribonucleic acid—to target, cut, and repair broken or damaged strands of DNA with new material.DNA, or deoxyribonucleic acid, conveys all hereditary data in people and different organic entities.
Researchers at Case Western Reserve University focused on controlling RNA, a polymer capable of deciphering genetic data and directing quality movement.RNA atoms can have different compound changes with indistinguishable hereditary groupings, however, and various properties and capacities.
By joining CRISPR with their synthetic interaction, the researchers oversaw the exact area and timing of changes to the RNA.
That progress appears to be fundamental for understanding and controlling the functions of these various RNA variants in major natural cycles and diseases, according to Liang.
All the more explicitly, Liang and his group utilized abscisic corrosive (ABA), a typical plant chemical, and a cycle called “synthetically instigated closeness.” By the same token, they were able to “state” or “eradicate” a primary component called m6A on a specific region of the RNA. Thus, they could really switch between two variants of RNA.
Researchers accept that m6A adjustment manages principal RNA cycles and properties but, at the same time, is straightforwardly connected to different human infections, he said.
The examination group likewise made a method for utilizing bright light to switch ABA “on,” making the altering system significantly more exact, Liang said.
“It makes the examination considerably more interesting that we accept it tends to be applied to other RNA changes, not simply m6A,” Liang said, “and the elements of the larger part of those alterations are absolutely obscure.”