Where and which human tissues contain which cell types? Which qualities are dynamic in the singular cells, and which proteins are tracked down there? A specialized atlas will provide answers to these and other questions, including how different tissues form during embryonic development and what causes diseases.
Researchers hope to map organoids, as well as tissue that has been isolated directly from humans, in this atlas. These are three-layered bunches of tissue that are developed in the research facility and created in a way like human organs, yet on a limited scale.
“The benefit of organoids is that we can mediate in their turn of events and test dynamic substances on them, which permits us to more deeply study solid tissue as well as sicknesses,” makes sense of Barbara Treutlein, Teacher of Quantitative Formative Science at the Branch of Biosystems Science and Designing at ETH Zurich in Basel.
“The benefit of organoids is that we can manipulate their development and test active substances on them, allowing us to learn more about both healthy tissue and diseases,”
Barbara Treutlein, Professor of Quantitative Developmental Biology.
Treutlein has developed a method to collect and compile a great deal of information about organoids and their development in order to assist in the production of such an atlas. This method was developed in collaboration with researchers from the universities of Zurich and Basel. The stem-cell-derived organoids of the human retina were the subject of this study’s application.
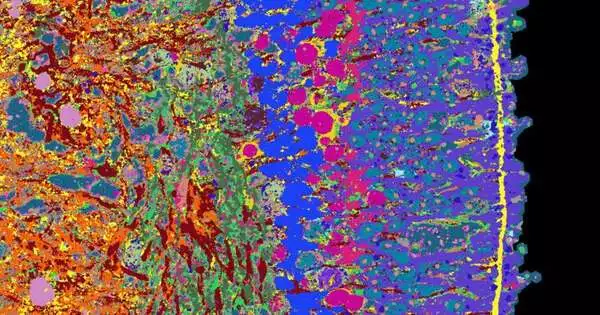
The 4i technology was at the heart of the methods that the scientists used to develop their strategy: iterative circuitous immunofluorescence imaging. Using fluorescence microscopy, this new imaging method can see several dozen proteins in a thin tissue section at high resolution. Lucas Pelkmans, a professor at the University of Zurich and co-author of the study that was recently published in Nature Biotechnology, developed the 4i technology a few years ago. The researchers used this approach for the first time with organoids in this study.
Typically, fluorescence microscopy is used by researchers to highlight three proteins in a tissue using distinct fluorescent dyes. For specialized reasons, staining in excess of five proteins all at once is beyond the realm of possibilities. Three dyes are used in 4i technology; however, after measurements have been taken from the tissue sample, these dyes are removed and three new proteins are stained. A robot carried out this step 18 times, and the entire procedure took 18 days.
In conclusion, a PC blends the singular pictures into a single microscopy picture on which 53 distinct proteins are noticeable. They provide information about how various cell types in the retina work together, for instance, ganglion cells, cones, and rods.
The specialists have enhanced this visual data on retinal proteins with data on which qualities are perused in the singular cells.
High spatial and temporal resolution The researchers conducted all of these analyses on organoids of varying ages and, as a result, of varying developmental stages. They were able to create a time series of images and genetic data that describes the development of retinal organoids over the course of 39 weeks.
“We can use this time series to demonstrate the organoid tissue’s gradual formation, the locations of the cell types that proliferate and when, and the synapses. The cycles are practically identical to those of retinal arrangement during an undeveloped turn of events,” says Dark Camp, a teacher at the College of Basel and a senior creator of this review.
The specialists distributed their picture data and more discoveries on retinal improvement on a freely accessible site called EyeSee4is.
In the future, the researchers hope to deliberately disrupt the development of retinal organoids using drugs or genetic modifications. “Up until now, the scientists have been studying how a healthy retina develops.” Camp claims that this will provide us with fresh insights into diseases like retinitis pigmentosa, a hereditary condition that causes the light-sensitive receptors in the retina to gradually degenerate, eventually leading to blindness. The analysts need to find out when this cycle starts and how it tends to be halted.
Treutlein and her partners are likewise chipping away at applying the new definite planning method to other tissue types, like various segments of the human cerebrum and different growth tissues. This will produce an atlas that details how human organoids and tissues develop step by step.
More information: Lucas Pelkmans, Multimodal spatiotemporal phenotyping of human retinal organoid development, Nature Biotechnology (2023). DOI: 10.1038/s41587-023-01747-2. www.nature.com/articles/s41587-023-01747-2