According to researchers at the University of Illinois at Urbana-Champaign, newly developed “smart” coatings for surgical orthopedic implants can kill infection-causing bacteria while simultaneously monitoring strain on the devices and providing early warning of implant failure. The coatings coordinate adaptable sensors with a nanostructured antibacterial surface triggered by the wings of dragonflies and cicadas.
In another concentrate in the journal Science Advances, a multidisciplinary group of specialists found the coatings forestalled disease in live mice and planned strain in business inserts applied to sheep spines to caution of different embedding or recuperating disappointments.
Qing Cao, a professor of materials science and engineering at the University of Illinois, was the study’s leader. “This is a combination of bio-inspired nanomaterial design with flexible electronics to battle a complicated, long-term biomedical problem,” he said.
“Using a mechanical approach to killing bacteria allowed us to avoid many of the problems associated with chemical approaches while still giving us the flexibility needed to apply the coating to implant surfaces,”
Pathobiology professor Gee Lau, a coauthor of the study.
According to Cao, the most common issues with orthopedic implants are infection and device failure, which affect up to 10% of patients. A few ways to deal with battling disease have been attempted; however, all have serious limits, he said. On surfaces that repel water, biofilms can still form, and coatings that are loaded with antibiotic chemicals or drugs run out in a matter of months. These coatings have toxic effects on the surrounding tissue and are ineffective against drug-resistant bacterial pathogens.
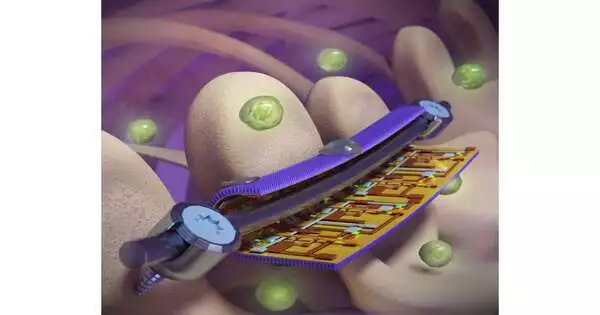
Taking inspiration from the normally antibacterial wings of cicadas and dragonflies, the Illinois group made a slender foil designed with nanoscale points of support like those tracked down on the bugs’ wings. The pillars puncture the cell wall of a bacterial cell when it tries to bind to the foil, killing it.

The gadgets join bug-propelled nanostructured foils with exceptionally delicate, adaptable hardware. Here the gadget models are applied to industrially accessible spinal embeds and shown on a spine model. Credit: Yi Zhang.
A professor of pathobiology and a coauthor of the study Gee Lau said, “Using a mechanical approach to killing bacteria allowed us to bypass a lot of the problems with chemical approaches while still giving us the flexibility needed to apply the coating to implant surfaces.”
For strain monitoring, the researchers integrated arrays of highly sensitive, flexible electronic sensors onto the back side of the nanostructured foil, where it comes into contact with the implant device. This could assist doctors with watching the recuperating progress of individual patients, guiding their restoration to abbreviate the recuperation time and limit dangers, and fixing or supplanting gadgets before they hit the weak spot, the scientists said.
The designing group then collaborated with veterinary clinical medication teacher Annette McCoy to test their model gadgets. They embedded the foils in live mice and observed them for any indication of contamination in any event when microorganisms were presented. They also applied the coatings to spinal implants that are already on the market and monitored the strain that the implants took under normal load on sheep spines in order to diagnose device failure. Both functions were satisfied by the coatings.
Cao stated that although the prototype electronics required wires, the researchers plan to develop wireless power and data communications interfaces for their coatings as the next step, which is essential for clinical application. They are likewise attempting to foster the huge-scale creation of the nanopillar-finished microorganism-killing foil.
Cao stated, “These types of antibacterial coatings have a lot of potential applications, and since ours uses a mechanical mechanism, it has potential for places where chemicals or heavy metal ions—as are used in commercial antimicrobial coatings currently—would be detrimental.” Cao was referring to the coatings that are currently available on the market.
More information: Yi Zhang et al, A smart coating with integrated physical antimicrobial and strain-mapping functionalities for orthopedic implants, Science Advances (2023). DOI: 10.1126/sciadv.adg7397