In a world that is gradually reducing most, if not all, connections with carbon-based energy, there has been a fleeting ascent in the utilization of lithium-particle batteries as a cutting-edge energy capacity arrangement. However, this has led to yet another issue: more lithium battery waste has been generated.
Over 500 charging and discharging cycles, lithium-ion batteries gradually degrade, losing anywhere from 12 to 24 percent of their total capacity. The electrolyte and different materials inside the battery can likewise corrupt, causing a decline in capacity over the long run. Due to the potential for toxic elements to leach into the soil and water, the disposal of lithium batteries in landfills or incineration can pose environmental and safety concerns.
Reusing lithium batteries requires broad utilization of hydrometallurgy, a part of metallurgy that includes watery arrangements. In a nutshell, the used battery is dismantled, valuable metals are extracted with solvent, and stripping is followed by the recovery of the extracted metal and the recycling of solvent. The extraction-stripping processes that are currently used for recycling lithium batteries have multiple steps, each of which necessitates its own reactor and has its own set of parameters. As a result, the recycling rate for lithium batteries is very low because this significantly increases the difficulty and cost of recycling.
“The technology is particularly cutting-edge in that, as we demonstrate, it can be tailored to work with feed metal compositions other than those used in batteries.”
Professor Bartosz A. Grzybowski at the Center of Soft and Living Matter within the Institute for Basic Science (IBS).
There have been numerous attempts to partition the reactor with membranes to create a one-pot, one-step solution. However, membrane failures, particularly when subjected to vigorous stirring, have ruled out these concepts in larger reactors.
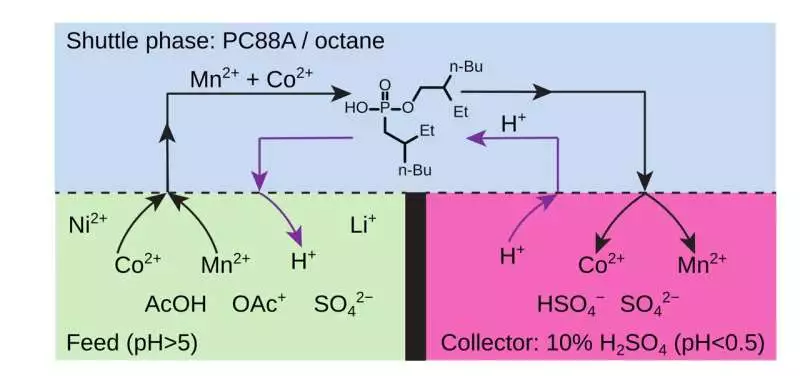
Figure 2. Schema for separating Mn (II), Co (II), and Li (I) from Ni (II) and Li (I) Credit: Institute for Basic Science
A novel method for recycling valuable metals like lithium, nickel, and cobalt from spent lithium-ion batteries was reported by an interdisciplinary research group led by Professor Bartosz A. Grzybowski at the Center of Soft and Living Matter within the Institute for Basic Science (IBS), South Korea, to address this issue.
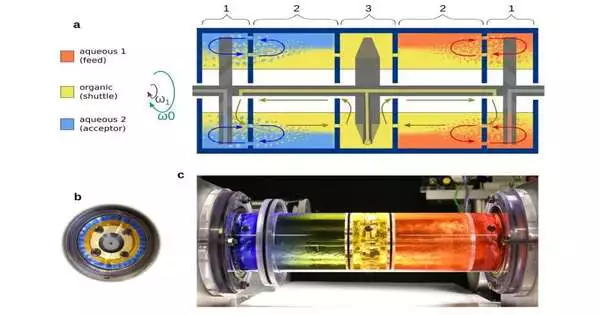
Gryzbowski’s group’s spinning concentric liquid reactors, which were found to be effective at carrying out multiple steps in a single chamber, have earned them fame in the field. This time, the group was successful in implementing this idea to simplify the lithium battery recycling extraction-stripping procedure. The study has been published in the Advanced Materials journal.
The on-a-plane turning reactor, which was planned by co-creator Dr. Olgierd Cybulski, can deal with complex metal combinations in which watery feed, natural extractant, and fluid acceptor stages are available in the equivalent pivoting vessel. This reactor can be vigorously stirred and emulsified without the coalescence of aqueous layers, in contrast to one-pot setups that use membranes. By placing all of these liquids in a rotating vessel so that they form concentric layers stable enough to allow efficient interfacial mixing without coalescing the aqueous layers, the arrangement of the higher-pH feed, organic extractant (shuttle), and lower-pH acceptor phases is robustly maintained.
According to co-author Dr. Cristóbal Quintana, this method “can perform the separation of metals in a matter of minutes, using a low concentration of extracting agents and with high selectivity,” which is quite impressive.

3. Figure 3: Pictures representing the advancement of the division interaction The feed phase changes color from dirty green to light green, indicating increased nickel purity. Credit: Institute for Basic Science
This study demonstrates that segmented concentric liquid reactors, in particular, can quickly separate valuable metals from highly concentrated mixtures using much lower extractant concentrations than conventional methods do and can access previously unknown process parameter ranges.
Concentric liquid reactors are an intriguing alternative to traditional hydrometallurgical methods and could potentially be utilized for the separation of other valuable metals due to their favorable power-to-operate versus reactor-size scaling. “The technology is also forward-looking in the sense that, as we show, it is tunable to different feed metal compositions and, obviously, to metals other than those used in batteries,” Professor Grzybowski explains.
More information: Cristóbal Quintana et al, One‐Pot, Three‐Phase Recycling of Metals from Li‐Ion Batteries in Rotating, Concentric‐Liquid Reactors, Advanced Materials (2023). DOI: 10.1002/adma.202211946