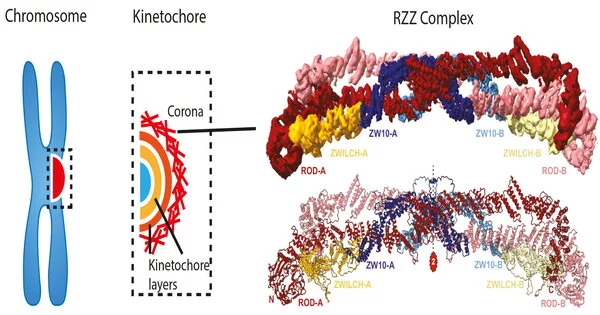
The 23 chromosomes that carry the human genome must be copied and then transferred to two newly developing daughter cells during cell division in the mother cell. The result, at least in healthy cells, is stunningly faultless, with no chromosome ever lost. Not so with cancer cells, where chromosome segregation mistakes produce a steady stream of novel genetic variations that promote metastatic growth and resistance to chemotherapy. The chromosomal delivery program is carried out by a multilayered protein structure known as the kinetochore. In a highly interdisciplinary joint tour de force, the Max Planck Institute of Molecular Physiology’s Andrea Musacchio and Stefan Raunser groups researched the kinetochore corona, the structure’s outermost layer. They discovered the structural organization of the corona’s key building block, the RZZ complex, and decoded the mechanism of corona construction using single particle cryo-electron imaging and protein reconstitution. Their findings shed light on the molecular underpinnings of genome inheritance across generations.
Cell division is the process by which our bodies are built, supplying all the cells in our tissues and organs, from the skin to the colon, and from the blood to the brain. It not only helps these organs to grow, but it also allows them to rejuvenate with new cells as necessary. Cell division begins with the replication of chromosomes, which are the bearers of the human genome’s three billion nucleotides. Mitosis is the process through which replicated chromosomes are transferred to daughter cells. The mitotic spindle, a network of thread-like structures, initially captures the chromosomes during mitosis. The spindle splits the chromosomes in opposite directions after arranging them in a finely choreographed procedure, so that when two cells arise from one, each inherits a perfect copy of the genome. Even little mistakes in this process will have disastrous physiological implications.
A multifaceted problem
The kinetochore is the place at which chromosomes make contact with the spindle, and it is thus significantly engaged in the process of chromosome alignment and partition. It is a multilayered, sophisticated protein complex. “Understanding kinetochores is a big difficulty since they are made up of numerous levels, each of which is made up of many interacting building blocks,” Musacchio explains. “The corona, the kinetochore’s outermost layer, has preserved some of the kinetochore’s most intriguing mysteries. Its construction is especially intriguing because the complex has a short lifetime that ends just before the essential processes of chromosomal alignment and segregation.
Musacchio’s laboratory previously made significant advances in understanding the structure and function of the several layers of kinetochores, as well as how they bind chromosomes to microtubules. The group used a reductionist strategy known as biochemical reconstitution to get this knowledge. They created the constituent components of the protein networks in a test tube, outside of the cell. They then pieced them back together to produce an almost full kinetochore that they could analyze in isolation, in a controlled and simple environment that contrasted with the extremely intricate, bustling interior of a cell.
“The outermost layer, the corona, has retained some of the most interesting secrets of the kinetochore. Its assembly is particularly interesting, since the complex has a brief lifetime that ends right before the critical steps of chromosome alignment and segregation.”
Musacchio
The skilled team of two postdocs, Tobias Raisch and Giuseppe Ciossani, two Ph.D. students, Ennio d’Amico and Verena Cmentowski, and other coworkers, was able to recreate the kinetochore corona using the same technique. They discovered that only two components are required: the ROD-Zwilch-ZW10 (RZZ) protein complex and the protein Spindly, which is required for the kinetochore’s interaction with the microtubules. The corona only forms on kinetochores, and the processes that limit its growth to these structures have remained an important unanswered subject. The scientists were able to identify an enzyme, the kinase MPS1, as the crucial catalyst of RZZ corona construction at the kinetochore by reconstituting the process in vitro.
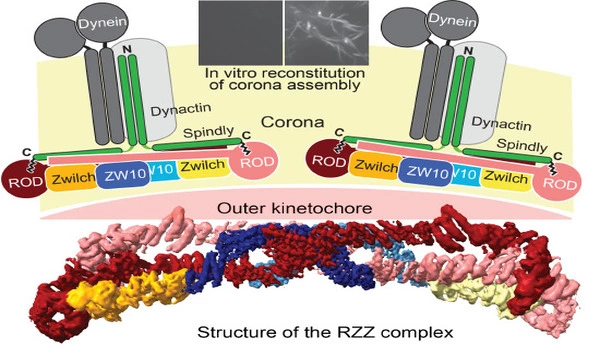
One step closer to the crown
Since the 1960s, electron microscopy (EM) has accompanied the study of kinetochores, but it wasn’t until recently that booming methodological improvements enabled this technology to view the building blocks at atomic size. “We created the first 3D structural model of the RZZ complex using cryo-EM in 2017,” explains Raunser. However, at this early model’s 1 nm resolution, it was unable to discern the minute molecular features responsible for biological activity.
The new structural study increased the resolution to the point where atomic features could be revealed, finally revealing how interactions between RZZ components and Spindly drive corona assembly into a vast polymer that surrounds the kinetochore. “Our discovery culminates a series of prior studies on the kinetochore corona, thus providing us with a framework to comprehend the key moment of cell division when chromosomal adhesion to microtubules becomes virtually irreversible,” Musacchio says. Future research will attempt to integrate the corona into reconstructed kinetochores, a significant step toward the in vitro restoration of chromosomal segregation, a lofty aim that will shed light on the fundamental mechanism of life.
The findings were published in the EMBO Journal.